Knee Embolization (GAE): An Emerging Treatment for Knee Arthritis Pain
Knee embolization (geniculate artery embolization) is an emerging minimally invasive procedure for knee osteoarthritis pain. Learn how GAE works, clinical evidence, costs, and who may benefit.
By Joint Pain Authority Team
Key Takeaways
- Geniculate artery embolization (GAE) is an emerging, minimally invasive procedure that reduces knee arthritis pain by blocking abnormal blood vessels in the inflamed joint lining
- Clinical trials show 50-70% pain reduction in many patients, with benefits lasting 12-24 months or longer
- The procedure takes about 60-90 minutes, uses local anesthesia, and patients typically go home the same day
- GAE is not FDA-approved for knee osteoarthritis — it is performed off-label by interventional radiologists using cleared embolic materials
- Cost ranges from $3,000-$15,000 depending on location and insurance; coverage is limited but expanding
- Best suited for patients with moderate knee OA who have not responded to conservative treatments and want to avoid or delay knee replacement surgery
If you have been dealing with knee osteoarthritis pain that has not improved with physical therapy, injections, or medications, you may have come across a newer option called knee embolization — formally known as geniculate artery embolization (GAE).
This procedure takes a completely different approach than traditional treatments. Instead of adding lubrication to the joint (like gel injections) or blocking pain signals (like nerve blocks), GAE targets the abnormal blood vessels that fuel inflammation inside your arthritic knee.
The research is still early, but the results so far are promising. Here is what you need to know.
What Is Knee Embolization (GAE)?
The Science Behind It
When osteoarthritis develops in your knee, the joint lining (called the synovium) becomes inflamed. This inflammation triggers the growth of abnormal new blood vessels — a process doctors call neovascularization. These abnormal vessels do two things that make your pain worse:
- They deliver more inflammatory chemicals to the joint, keeping the cycle of inflammation going
- They bring in new nerve fibers (neoinnervation) that make the area more sensitive to pain
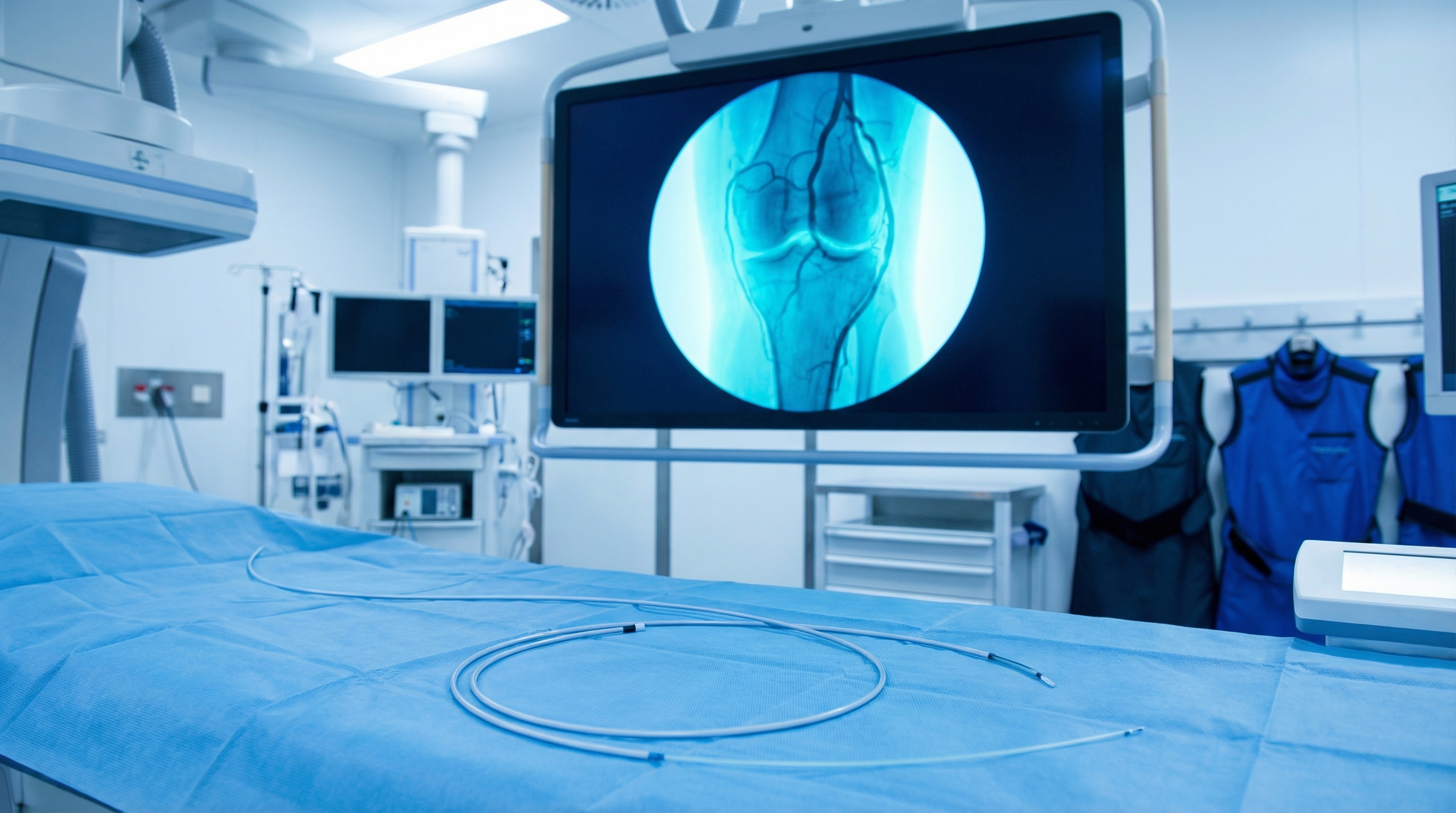
GAE works by selectively blocking these abnormal blood vessels. An interventional radiologist threads a tiny catheter through an artery — usually in the upper thigh — and guides it to the small arteries around your knee (called genicular arteries). Once there, they inject microscopic beads (called microspheres) that plug the abnormal vessels feeding the inflamed tissue.
By cutting off blood supply to the inflamed synovium, GAE reduces both the inflammation and the nerve sensitivity that cause your pain.
What GAE Does Not Do
It is important to understand what this procedure is not:
- It does not regrow cartilage or reverse arthritis damage
- It does not replace the joint like knee replacement surgery
- It does not block nerve signals like radiofrequency ablation
- It is not a cure for osteoarthritis
GAE is a pain management procedure that targets the inflammatory component of knee OA. Your arthritis is still there, but the painful inflammation may be significantly reduced.
How the Procedure Works
GAE is performed as an outpatient procedure — you go home the same day. Here is what to expect step by step.
Before the Procedure
- Your doctor reviews imaging (X-rays, possibly MRI) to confirm osteoarthritis and assess the level of joint inflammation
- Radiopaque markers may be placed on your skin at the points where you feel the most pain, helping guide the radiologist during the procedure
- You receive local anesthesia and light sedation (you are awake but relaxed)
During the Procedure (60-90 Minutes)
-
Arterial access: A small puncture is made in the upper thigh (femoral artery) or sometimes the wrist (radial artery). No surgical incision is needed.
-
Catheter navigation: Using real-time X-ray imaging (fluoroscopy), the radiologist guides a thin catheter and even thinner microcatheter into the genicular arteries around your knee.
-
Identifying abnormal vessels: Contrast dye is injected to reveal which arteries are feeding the inflamed synovial tissue. These show up as areas of abnormal “blush” on the imaging.
-
Embolization: Tiny microspheres (typically 100-300 micrometers, smaller than a grain of sand) are carefully injected to block the abnormal vessels. The radiologist stops when the inflammatory blush disappears while keeping the main artery open.
-
Completion: The catheter is removed, and a small bandage is placed over the puncture site.
After the Procedure
- You rest in recovery for a few hours while the sedation wears off
- Most patients go home 4-6 hours after the procedure
- You can walk immediately but should avoid strenuous activity for 1-2 weeks
- Mild knee soreness is normal for 3-5 days
Recovery timeline: Most patients notice meaningful pain improvement within 1-4 weeks. Maximum benefit typically develops over 2-6 months as the inflammation fully resolves. Many patients report that better sleep quality is one of the first improvements they notice.
Clinical Evidence: What Research Shows
Evidence level: Emerging. GAE has growing Level 1 evidence from randomized controlled trials, but studies remain relatively small. Larger trials are ongoing. This is a promising but still developing treatment.
Key Studies and Results
Several clinical trials have evaluated GAE for knee osteoarthritis pain. Here are the most important findings:
Bagla Randomized Controlled Trial (2022)
| Detail | Result |
|---|---|
| Study type | Multicenter double-blind RCT vs. sham procedure |
| Patients | 21 (14 GAE, 7 sham) |
| Pain reduction (GAE group) | 51-point VAS decrease at 1 month |
| Pain reduction (sham group) | 1-point VAS decrease at 1 month |
| Significance | p < 0.01 |
| Key finding | All 7 sham patients crossed over to GAE due to lack of improvement |
GENESIS Trial — 24-Month Follow-Up (2024)
| Detail | Result |
|---|---|
| Study type | Prospective single-center pilot (UK) |
| Patients | 46 (mostly KL Grade 3 OA) |
| Technical success | 87% |
| VAS pain reduction | ~36% improvement at 2 years |
| MRI findings | Significant reduction in synovitis; no osteonecrosis |
| Key finding | 9 patients later had knee replacement; GAE did not complicate surgery |
Meta-Analysis of 270 Patients (Taslakian, 2025)
| Detail | Result |
|---|---|
| Studies included | 9 studies, 270 patients, 339 knees |
| Technical success | 99.7% |
| Pain scale reduction | 34-39 points at 12 months |
| WOMAC improvement | 28-34 points total score improvement |
| Patients meeting MCID for pain | 78% at 12 months |
| Patients meeting MCID for function | 92% at 12 months |
| Knee replacement rate | Only 5.2% went on to TKA during follow-up |
What the Numbers Mean
Across studies, the general pattern shows:
- 50-70% of patients experience at least 50% pain reduction at 12-24 months
- 78% meet the “minimally clinically important difference” for pain improvement — meaning the improvement is large enough to notice in daily life
- 92% show meaningful functional improvement on standardized questionnaires
- Results are best in patients with moderate disease (Kellgren-Lawrence grades 2-3)
- Only about 5% of patients go on to need knee replacement during follow-up periods
Ongoing Trials to Watch
Several major trials will provide more data in the coming years:
- MOTION Trial: An international RCT comparing GAE to cortisone injections across the US, UK, Australia, Brazil, and Canada
- GENESIS II: A UK-based RCT comparing GAE to sham procedure
- GRAVITY Trial: Comparing GAE to conservative management (results expected 2027)
- RESORB Trial: The first US FDA pivotal trial testing a new resorbable microsphere (Nexsphere-F), which dissolves within hours rather than blocking vessels permanently
How GAE Compares to Other Treatments
| Feature | GAE | Cortisone Injections | HA Injections | Nerve Blocks/RFA | Knee Replacement |
|---|---|---|---|---|---|
| How it works | Blocks inflamed blood vessels | Reduces inflammation in joint | Lubricates joint | Disables pain nerves | Replaces joint |
| Duration of relief | 12-24+ months | 4-12 weeks | 3-6 months | 6-12 months | Permanent (15-20 yr implant life) |
| Procedure time | 60-90 min | 5-10 min | 5-10 min | 30-45 min | 1-2 hours |
| Anesthesia | Local + sedation | Local | None/local | Local | General |
| Recovery | 1-2 weeks light duty | Same day | Same day | 1-3 days | 6-12 weeks |
| Typical cost | $3,000-$15,000 | $100-$300 | $500-$1,500 | $2,000-$5,000 | $40,000-$80,000+ |
| Insurance coverage | Limited | Covered | Usually covered | Usually covered | Covered |
| Evidence level | Emerging | Strong | Moderate | Moderate | Strong |
| FDA status | Off-label | Approved | Approved | Approved | Approved |
Where GAE Fits In
GAE fills a specific gap in the treatment ladder. It may be a good option when:
- Cortisone injections provide only short-term relief or have stopped working
- HA injections are not providing enough benefit
- You are not ready for knee replacement or want to delay it
- You want longer-lasting relief than what injections typically provide
It is important to note that GAE is not meant to replace these proven treatments. For many patients, starting with conservative options — physical therapy, weight management, medications, and injections — remains the right first step.
Cost and Insurance Coverage
Current Cost Landscape
GAE costs vary significantly depending on where you have the procedure:
- Estimated total cost: $3,000-$15,000 (facility fees, radiologist fees, imaging, supplies)
- Most common range: $5,000-$10,000
Insurance Coverage (As of Early 2026)
Coverage for GAE is currently limited and variable:
Medicare (Traditional)
- Generally covers 80% of the procedure cost
- Patient responsibility: approximately $1,500-$2,500 after deductibles
- Medicare + Medigap supplement may result in minimal out-of-pocket costs
Medicare Advantage
- Coverage varies by plan
- Some plans have fixed copays (around $75)
- Prior authorization typically required
Commercial Insurance (Aetna, BCBS, Cigna, Humana, UnitedHealthcare)
- Most currently classify GAE as “investigational” or “experimental”
- Coverage denials are common
- Medical exceptions may be granted when patients are ineligible for surgery, have failed other treatments, or have medical conditions making surgery too risky
Important: Before scheduling GAE, contact your insurance provider to verify coverage. Ask specifically about CPT codes for geniculate artery embolization. Many GAE centers have insurance coordinators who can help navigate the authorization process and appeal denials.
Will Coverage Improve?
There are positive signs. One new resorbable microsphere (Nexsphere-F) has received FDA Breakthrough Device Designation and CMS IDE Category B approval, which provides Medicare coverage for patients enrolled in its clinical trial. As more trial data is published and FDA-approved devices reach the market, broader insurance coverage is likely to follow — though this process may take several years.
Who Is a Good Candidate?
Potentially Good Candidates
GAE may be worth discussing with your doctor if you:
- Have moderate knee osteoarthritis (Kellgren-Lawrence grades 2-3) confirmed on X-ray
- Experience persistent knee pain despite at least 3-6 months of conservative treatment
- Have tried physical therapy, medications, and/or injections without adequate relief
- Are not ready for knee replacement or want to delay surgery
- Are not a good surgical candidate due to other health conditions, obesity, or blood thinner use
- Have localized tenderness around the knee (suggesting active synovial inflammation)
Less Ideal Candidates
GAE may not be the best option if you:
- Have severe “bone-on-bone” arthritis (KL Grade 4) — results tend to be less durable
- Are a good candidate for knee replacement and ready for definitive treatment
- Have significant peripheral artery disease (the genicular arteries serve as important blood supply pathways)
- Have an active infection or malignancy near the knee
- Expect a complete cure for your arthritis
Age Range
Most clinical trials have enrolled patients aged 40-80. There is limited data on patients under 40 or over 80.
Risks and Limitations
Known Risks
Based on clinical trial data, GAE has a favorable safety profile, but risks include:
- Skin discoloration near the knee (most common; occurs in about 12% of patients, typically resolves within days)
- Minor bruising or soreness at the catheter insertion site
- Temporary numbness or tingling in the leg (rare, usually resolves within 2 weeks)
- Groin hematoma at the access site (rare)
- Deep vein thrombosis (very rare; one case reported in the GENESIS trial, attributed to patient immobilization)
What Has NOT Been Seen
Importantly, across published trials:
- No osteonecrosis (bone death) has been found on follow-up MRI scans
- No cartilage damage has been attributed to the procedure
- No serious systemic complications (infection, organ damage) have been reported
- GAE does not prevent future knee replacement if you eventually need one
Bridge to surgery: A multi-institution study of 47 patients who had knee replacement after prior GAE found that the complication rate was comparable to standard knee replacement. GAE does not “burn bridges” — you can still have surgery later if needed.
Key Limitations
- Long-term data is limited — most studies have 1-2 year follow-up, with some extending to 3-4 years
- Not all patients respond — about 20-30% may not achieve meaningful pain relief
- No standardized protocol yet — embolic materials, particle sizes, and techniques vary between centers
- Results may fade over time — some patients may need repeat treatment
- Study sizes are still small — the largest meta-analysis includes 270 patients across 9 studies
Where to Find GAE Providers
GAE is performed by interventional radiologists — doctors who specialize in image-guided, catheter-based procedures. Finding an experienced provider is important.
Where to Look
- Academic medical centers with interventional radiology departments (many leading hospitals now offer GAE)
- Specialized vascular and interventional radiology practices in major metro areas
- Clinical trial sites — enrolling in a trial may provide access to GAE with reduced or no cost
What to Ask a Potential Provider
- How many GAE procedures have you performed? (Look for providers with meaningful experience)
- What embolic material do you use? (Microspheres, imipenem/cilastatin, or resorbable beads each have different profiles)
- What outcomes have your patients experienced? (Ask for their own results data)
- Do you track your results systematically?
- What is the total cost, and will your office help with insurance?
Frequently Asked Questions
Is GAE FDA-approved?
No. As of early 2026, GAE is not specifically FDA-approved for knee osteoarthritis. The embolic materials used (microspheres) are FDA-cleared for other conditions (like uterine fibroids), but their use in knee OA is considered off-label. Multiple FDA-tracked clinical trials are underway, including the RESORB pivotal trial, which could lead to the first FDA-approved device specifically for this procedure.
How long does pain relief last after GAE?
Clinical studies show meaningful pain relief lasting 12-24 months in most responders. Some patients report benefits extending 3-4 years after a single procedure. However, since osteoarthritis is a progressive condition, pain may eventually return. The procedure can typically be repeated if needed.
Can I have GAE on both knees?
Yes, but typically one knee is treated at a time. Your doctor may recommend waiting several weeks between procedures. Some clinical protocols treat both knees in a single session, but this is less common.
Is GAE painful?
Most patients report minimal discomfort during the procedure thanks to local anesthesia and sedation. You may feel pressure or warmth in your knee during the embolization. Post-procedure soreness is typically mild and manageable with over-the-counter pain medication for 3-5 days.
How does GAE compare to PRP injections?
Both are relatively newer approaches for knee OA. PRP injections use concentrated platelets from your own blood to promote healing. GAE targets the inflammatory blood vessels directly. PRP effects typically last 6-12 months, while GAE may provide 12-24 months of relief. They target different aspects of osteoarthritis and could potentially complement each other, though no studies have evaluated combining them.
What if GAE does not work for me?
If GAE does not provide adequate relief, all other treatment options remain available. You can still pursue cortisone injections, hyaluronic acid injections, physical therapy, or knee replacement surgery. GAE does not limit your future options.
Stay ahead of joint pain
Get weekly insights on joint health and pain relief delivered to your inbox.
Join 10,000+ readers. No spam.
Related Resources
- Knee Osteoarthritis: Complete Guide — Understanding your condition
- Non-Surgical Knee Arthritis Treatments — All your options before surgery
- Regenerative Medicine for Joint Pain — PRP, stem cells, and emerging therapies
- Three Types of Knee Injections Compared — Cortisone, HA, and PRP
- Hyaluronic Acid Injections — How gel shots work
References
- Bagla S, Piechowiak R, Hartman T, et al. Genicular artery embolization for the treatment of knee pain secondary to osteoarthritis. J Vasc Interv Radiol. 2022;33(1):2-9.
- Little M, et al. GENESIS trial: Genicular artery embolization for knee osteoarthritis — 24-month outcomes. J Vasc Interv Radiol. 2024.
- Taslakian B, et al. Meta-analysis of genicular artery embolization for knee osteoarthritis: safety and efficacy across 270 patients and 339 knees. Osteoarthritis Cartilage Open. 2025.
- Landers S, et al. Genicular artery embolization for early-to-moderate knee osteoarthritis: randomized controlled trial. Radiology. 2023;307(2).
- MOTION trial. Comparison of genicular artery embolization vs intra-articular corticosteroid injection (NCT05818150). ClinicalTrials.gov.
- NEXTBIOMEDICAL. First patient enrolled in US FDA pivotal trial of Nexsphere-F for knee osteoarthritis pain (RESORB trial). Press release, October 2025.
- Ashraf A, et al. Systematic review of safety and efficacy of GAE for managing knee osteoarthritis. 2025.
- Okuno Y, et al. Transcatheter arterial embolization as a treatment for medial knee pain in patients with mild to moderate osteoarthritis. Cardiovasc Intervent Radiol. 2015;38(2):336-343.
Last reviewed: March 2026
Medical Disclaimer: This article is for informational purposes only and does not constitute medical advice. Geniculate artery embolization is an emerging procedure that is not FDA-approved for knee osteoarthritis. Evidence is promising but limited to relatively small clinical trials with short-to-medium-term follow-up. Discuss all treatment options with your healthcare provider. Individual results vary, and not all patients will experience the outcomes described in clinical studies.
Topics
Enjoyed this article?
Get more insights like this delivered to your inbox weekly.
Join 10,000+ readers. No spam.